 
Where will the largest jump in ionization energies occur for aluminum? Between the third and fourth ionization energies For the outermost electron of an atom, effective nuclear charge as you go from left to right on the periodic table. Each ionization energy increases for successive electrons being removed. Which of the following elements has the largest atomic radius? Rubidium, Rb Elements have a series of ionization energies for removing the first and additional electrons from the atom. Cations are smaller than their neutral parent atoms because there are fewer electrons "competing" for the attractive force of the protons in the nucleus. Which element is the most metallic? Cs Select the true statement. 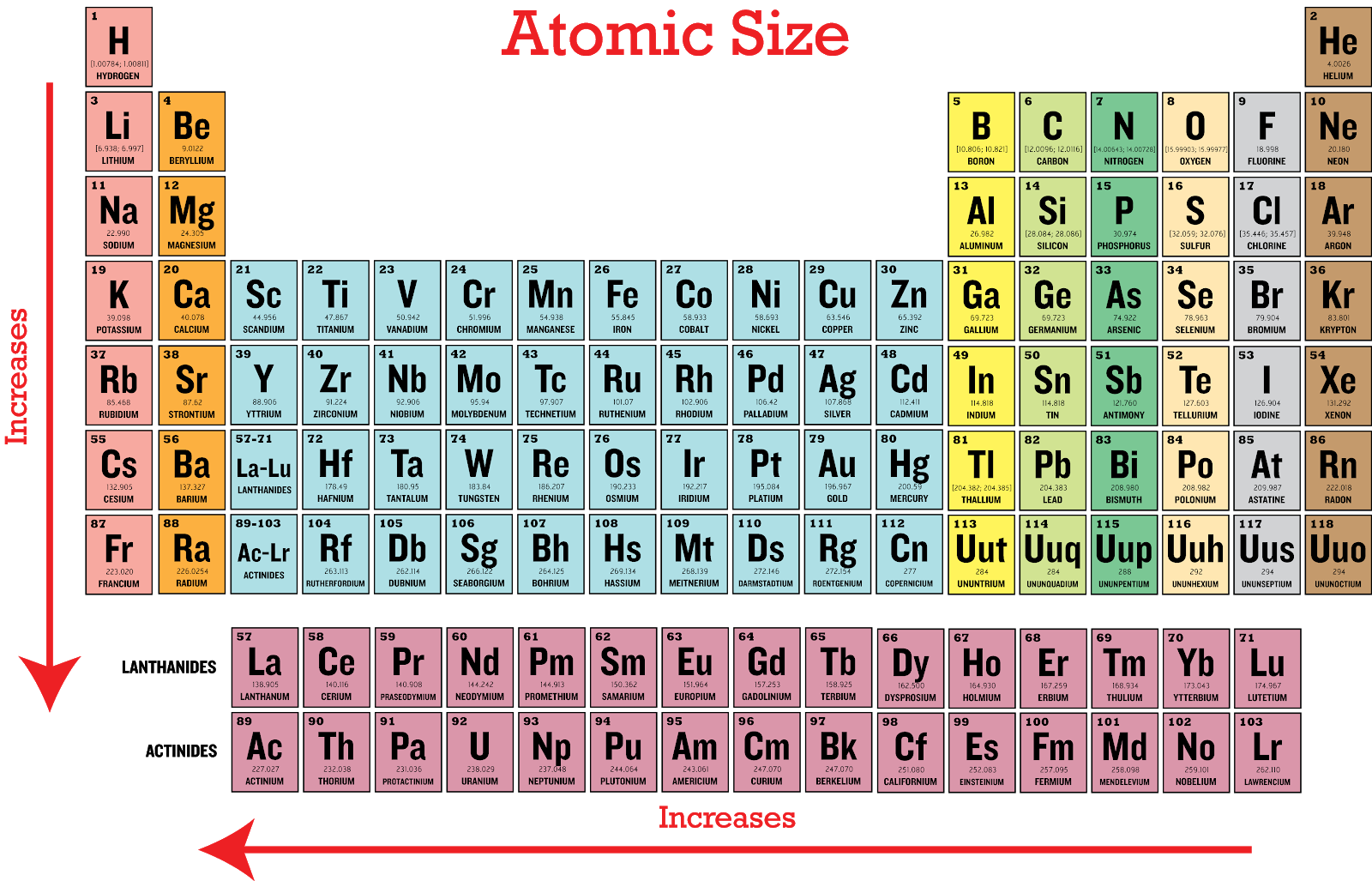
larger smaller Which element requires the most energy to lose one electron? Helium Which one of the following statements is true regarding ionization energies? Ionization energies are all endothermic. Which element could have the following series of ionization energies (IE)? Magnesium Which pair lists the element with the lower first ionization energy first? Oxygen, fluorine Anions are _ than their neutral parent atom, and cations are _ than their neutral parent atom. decreases more positive Additional energy is required to move second and successive electrons from an ion. Moving from lithium to cesium, the melting point _ and the electron affinity becomes _. Which element will react with these noble gases to form compounds? Fluorine Which sequence lists the ions in order of increasing radius (smallest to largest)? Mg2+ < Na+ < F− < O2− Alkali metals show trends, such as electron affinity and melting point, that are not typically seen in other groups of metals. However, a few compounds are known to form with krypton and xenon. Noble gases are named as such because they are very unreactive. Cl− Why are the electron affinity values for the noble gases endothermic? An electron added to a noble gas must go in the next higher energy level.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
